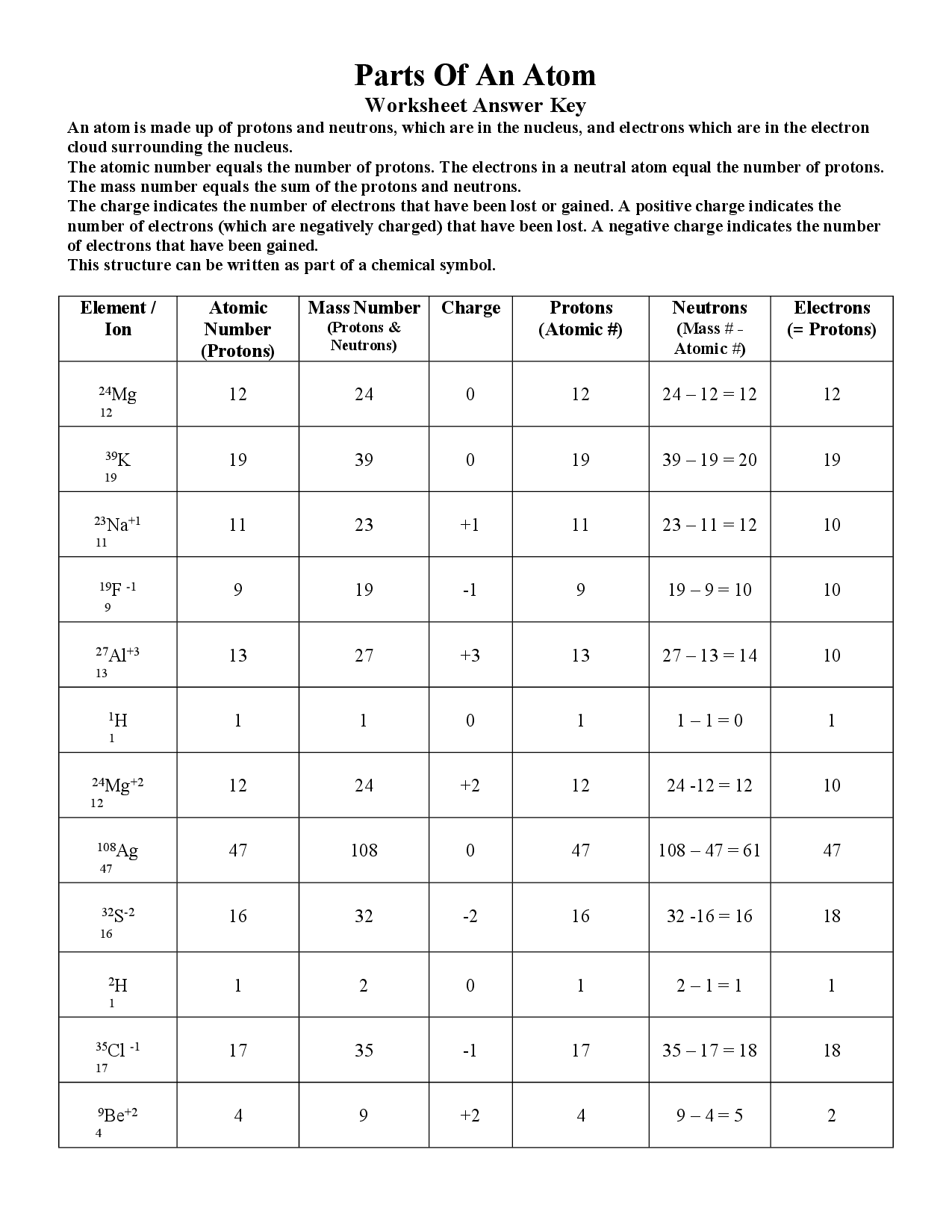
Have students use the internet to do this “Chemistry Scavenger Hunt”. Grade 6 elements, compounds and mixtures. Cathode ray tube is the guts of the oscilloscope and it generates the electron bean, accelerates the beam and deflects the beam. Cathode Ray Tube – The Cathode Ray Experiment by J.J.Thomson helped to find electrons. Obtain fast suggestions and results to know how nicely you carried out. Some of the worksheets for this concept are protons neutrons and electrons practice work reply key, construction of matter work solutions key ebook, atomic construction work 1 solutions, atomic construction evaluate.īest Pictures Of Bohr Mannequin Worksheet The atomic quantity provides the identity of a component in addition to its location on the periodic table. The cost is written on the top right facet.Ītomic and molecular construction a. No two totally different parts could have the. If you don’t mind share your remark with us and our followers at remark field at the bottom web page, additionally, do not forget to. These worksheets have college students discover the character of atoms and their structure. 2) Atoms of one factor are identical whereas atoms of … Pudding Model, 1900. 1) Elements are manufactured from tiny particles referred to as atoms. 3.0.4 The Pythagorean Theorem Worksheetĭalton’s Atomic Theory.3.0.3 Calculating Average Atomic Mass Worksheet.3 Related posts of "Worksheet Atomic Structure Answers".2.2 Obtain Atomic Structure Apply Worksheet Solutions:.2 Subatomic Particles And Isotopes Worksheets Solutions.1.1 Best Pictures Of Bohr Mannequin Worksheet.And finally, the charge of an atom is determined by the number of protons and electrons in the atom. The mass number of an atom is found by adding together the number of protons and neutrons. The number of electrons will determine how atoms interact with one another and decide if the atom as a whole is positive, negative, or neutral. The charge of an atom is calculated based on the difference between the number of protons in the nucleus and the number of electrons orbiting the nucleus.Īn atom is made up of protons, electrons, and neutrons. The periodic table arranges atoms in increasing atomic number. Atoms have an atomic number, which is the number of protons in the nucleus. Now that we have talked about the different parts of the atom, let’s summarize a few properties of atoms. Water is made up of two hydrogen atoms and one oxygen atom. Atoms can share electrons to form molecules, which are particles made up of many atoms. The electrons determine how atoms interact with each other. With fewer electrons than protons, the atom will have a positive charge. If the atom has more electrons than protons, its charge will be negative. If the number of electrons is equal to the number of protons, the overall electric charge of the atom is neutral. The electrons are attracted to the positive nucleus, but they can escape their orbit by an outside force.Ītoms have a certain number of electrons orbiting the nucleus. Orbiting the atom’s positively charged center are particles with a negative charge called electrons. This gives us names like Carbon-12 or Carbon-14, which are types of carbon atoms used in carbon dating! To find the mass number of an element you add the number of protons and neutrons together, so protons + neutrons = mass number. Usually, a stable atom has an equal number of neutrons and protons, but there are exceptions. In general, an atom will have a specific number of neutrons in the nucleus, meaning the atom won’t lose or gain any neutrons for a very long time. For example, hydrogen is an element with one proton in the nucleus and carbon is an element with 6 protons. A group of atoms that all have the exact same number of protons is called an element. Protons are very small, positively-charged particles, and neutrons are neutral particles that have no charge.Ītoms can have just one proton or they can have multiple. What is an atom?Īt the very center of an atom is the nucleus, which is made up of small particles called protons and neutrons. 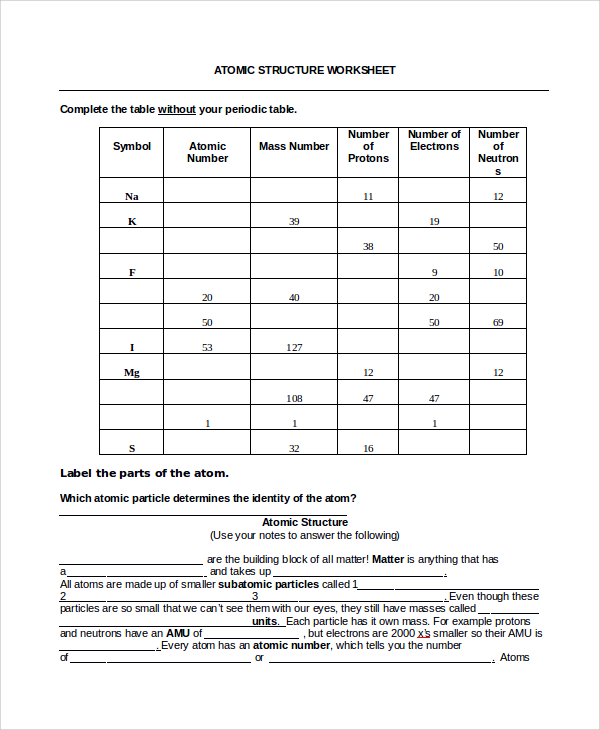
Atoms are the building blocks of the universe they make up everything you see around you.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
